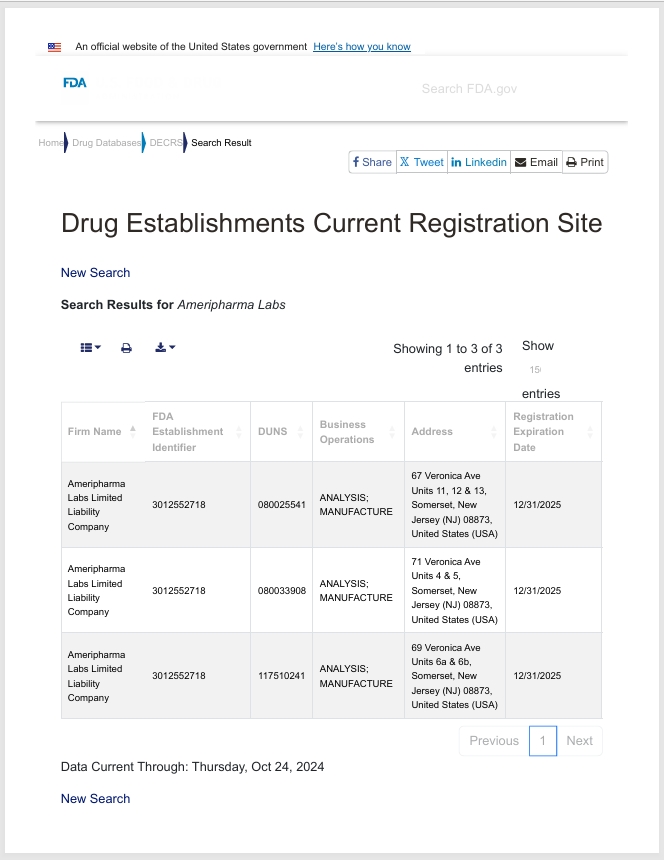
-
38,200 Sq Ft
-
Experts in large batch manufacturing
-
Product Development & Tech-transfers
-
In-house Lab testing and stability
-
Manufacturing capabilities
- Granulation
- Direct Blending
- Tablet Compression
- Tablet Coating


















.png)


.png)